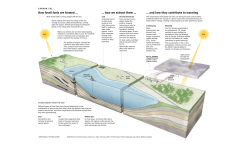
MOXUS Metabolic System Instruction Manual 1
MOXUS Metabolic System Instruction Manual 1 CONTENTS PAGE WARRANTY CAUTIONS 4 5 SYSTEM SETUP 6 SOFTWARE SETUP 7 DAILY SYSTEM STARTUP & SHUTDOWN 10 SYSTEM CALIBRATION Gas Calibration Gas Verification Volume Calibration Volume Verification Calibrating Other Channels 12 12 14 15 17 17 CONDUCTING A TEST Subject Information Test Preparation Start Test End Test 18 18 19 19 20 RUN TIME FEATURES Data Graphs Tabular Display Export to Excel Edit Data Hide/Unhide Column Column Move From/Move To Sort Column Ascending/Descending Select Column Properties Mark Invalid/Valid Data Column Width Averaging Interval 21 21 22 22 23 23 23 24 24 24 25 25 25 OTHER FEATURES 27 REPORTS 29 2 CONTENTS CONT’D. PAGE APPENDIX A – THEORY OF OPERATION & FORMULAS 31 APPENDIX B – COMPUTER REQUIREMENTS 42 APPENDIX C – CONSUMABLES / SPARE PARTS 43 APPENDIX D - TROUBLESHOOTING 45 APPENDIX E – CARDIAC OUTPUT MODULE [optional] 46 APPENDIX F – CANOPY SYSTEM [optional] 61 APPENDIX G – CLINICAL SYSTEM OPERATION 63 3 Warranty and Claims We warrant that any equipment of our own manufacture or manufactured for us pursuant to our specifications which shall not be, at the time of shipment thereof by or for us, free from defects in material or workmanship under normal use and service will be repaired or replaced (at our option) by us free of charge, provided that written notice of such defect is received by us within eighteen (18) months from date of shipment. All equipment requiring repair or replacement under this warranty shall be returned to us at our factory, or at such other location as we may designate, transportation prepaid. We shall examine such returned equipment and if it is found to be defective as a result of defective materials or workmanship, it shall be repaired or replaced as aforesaid. Our obligation does not include the cost of furnishing any labor in connection with the installation of such repaired or replaced equipment or parts thereof, nor does it include the responsibility or cost of transportation. In addition, instead of repairing or replacing the equipment returned to us as aforesaid, we may, at our option, take back the defective equipment and refund in full settlement the purchase price thereof paid to Buyer. If you are returning equipment from outside the United States, a statement should appear on the documentation accompanying the equipment being returned declaring that the goods being returned for repair are American goods, the name of the firm who purchased the goods, and the shipment date. This warranty shall not apply to any equipment (or part thereof) which has been tampered with or altered after leaving our control, or which has been repaired by anyone except us, or which has been subject to misuse, neglect, abuse, or improper use. Misuse or abuse of the equipment, or any part thereof, shall be construed to include, but shall not be limited to, damage by negligence, accident, fire, or force of the elements. Improper use or misapplication shall be construed to include improper or inadequate protection against shock, vibration, high or low temperature, overpressure, excess voltage and the like, or operating the equipment with or in a corrosive, explosive or combustible medium unless the equipment is specifically designed for such service, or exposure to any other service or environment of greater severity than that for which the equipment was designed. This warranty does not apply to used or second-hand equipment, nor extend to anyone other than the origina1 purchaser from us. THIS WARRANTY IS GIVEN AND ACCEPTED IN LIEU OF ALL OTHER WARRANTIES, WHETHER EXPRESS OR IMPLIED, INCLUDING WITHOUT LIMITATION ANY WARRANTIES OF FITNESS OR OF MERCHANTABILITY OTHER THAN AS EXPRESSLY SET FORTH HEREIN, AND OF ALL OTHER OBLIGATIONS OR LIABILIES ON OUR PART. IN NO EVENT SHALL WE BE LIABLE UNDER THIS WARRANTY OR ANY OTHER PROVISION OF THIS AGREEMENT FOR ANY ANTICIPATED OR LOST PROFITS, INCIDENTAL DAMAGES, CONSEQUENTIAL DAMAGES, TIME CHARGES, OR ANY OTHER LOSSES INCURRED BY THE ORIGINAL PURCHASER OR ANY THIRD PARTY IN CONNECTION WITH THE PURCHASE, INSTALLATION, REPAIR, OR OPERATION OF EQUIPMENT, OR ANY PART THEREOF, COVERED BY THIS WARRANTY OR OTHERWISE, WE MAKE NO WARRANTY, EXPRESS, OR IMPLIED, INCLUDING WITHOUT LIMITATION ANY WARRANTIES OF FITNESS OR OF MERCHANTABILITY, AS TO ANY OTHER MANUFACTURER’S EQUIPMENT, WHETHER SOLD SEPARATELY OR IN CONJUNCTION WITH THE EQUIPMENT OF OUR MANUFACTURE. WE DO NOT AUTHORIZE ANY REPRESENTATIVE OR OTHER PERSON TO ASSUME FOR US ANY OTHER LIABILITY IN CONNECTION WITH EQUIPMENT, OR ANY PART THEREOF, COVERED BY THIS WARRANTY. 4 ***CAUTION*** DO NOT USE THIS INSTRUMENT IN THE PRESENCE OF FLAMMABLE ANESTHETICS OR FLUORINATED GASES. NOT FOR USE WITH ANESTHETIC GAS MIXTURES. THESE INSTRUMENTS MUST BE ELECTRICALLY GROUNDED. DO NOT USE AN UNGROUNDED CONNECTOR OF ANY KIND. DO NOT OPEN CABINETS WITHOUT PRIOR FACTORY AUTHORIZATION. FOR HIGHEST ACCURACY, TOP COVER MUST REMAIN ON INSTRUMENTS. 5 SYSTEM SETUP FOR INSTALLATION, SETUP AND CHECKOUT OF SYSTEM COMPONENTS REFER TO THE INSTALLATION GUIDE INCLUDED WITH THE SYSTEM. ALSO REFER TO GAS ANALYZER MANUALS AND CLEANING INSTRUCTIONS. PLEASE REVIEW APPENDIX A: AEI METABOLIC SYSTEM THEORY OF OPERATION & FORMULAS, FOR A SYSTEM OVERVIEW AND DETAILS NECESSARY FOR CORRECT OPERATION OF THE SYSTEM AND INTERPRETATION OF THE DATA. ALL AEI METABOLIC SYSTEM MANUALS, INCLUDING THE INSTALLATION GUIDE AND THIS MANUAL, ARE LOCATED ON THE SOFTWARE CD AND ARE ALSO INSTALLED IN THE C:\PROGRAM FILES\AEI TECHNOLOGIES DIRECTORY ON THE COMPUTER. IT IS RECCOMMENDED THAT BOTH THE S-3A O2 ANALYZER AND CD-3A CO2 ANALYZER ALWAYS REMAIN POWERED ON. THIS WILL GREATLY REDUCE SYSTEM WARM-UP TIME AND PROLONG THE LIFE OF THE ANALYZERS. THE R-1 PUMP, INTERFACE BOX, COMPUTER AND PRINTER MAY BE TURNED OFF WHEN NOT IN USE. NOTE: THE MOXUS SYSTEM IS NOW ON THE 3RD GENERATION. THE FIRST GENERATION MOXUS HAD THE DATA ACQUISITION ELECTRONICS INSTALLED ON THE COMPUTER AND RAN UNDER THE DOS OPERATING SYSTEM. THE 2ND GENERATION MOXUS DATA ACQUISITION ELECTRONICS WERE PLACED IN A LONG ALUMINUM ENCLOSURE [INTERFACE BOX]. THE 3RD GENERATION MOXUS DATA ACQUISITION ELECTRONICS ARE PLACED IN A SQUARE PLASTIC BOX [INTERFACE BOX]. THIS MANUAL IS FOR THE 3RD GENERATION MOXUS ONLY. FOR PREVIOUS GENERATIONS PLEASE REFER TO THE ORIGINAL MANUAL PROVIDED WITH THE SYSTEM. 6 SOFTWARE SETUP Run the AEI Metabolic System Software program by double-clicking the icon on your desktop. A box of text in the lower right corner of the screen asks you to Please Wait. Wait until this box disappears. When the software has been loaded properly the box will disappear and the software is ready for use. NOTE: Do not attempt to operate the software until the Please Wait message disappears. Operating Modes and Options IMPORTANT! Required for proper operation • • • • • • • • • • • • • In the Menu Select: Tools > Hardware Dx. Select System Mode as ‘MOXUS’. Select Volume Source as ‘Pneumotach’ [or ‘Turbine [VMM-400]’ or ‘Turbine [KTC3]’ on some models, if used]. De-Select ‘Calibration gas = High/Low” if checked [unless otherwise advised]. For Cardiac Output Option see Appendix E. For Canopy System Option see Appendix F. For Clinical System see Appendix G. For automatic inspiratory fiO2 and fiCO2 sampling [Canopy System Option only] click ‘Sample Inspiratory Air’ The Canopy System Option can allow for automatically sampling the inspiratory O2 and CO2 concentrations every 10 minutes. Ten minutes after the Test has started collecting data the system will automatically switch to start sampling from the Sample Air input instead of the mixing chamber; with the data being marked Invalid. After 30 seconds the system will automatically switch back to start sampling from the mixing chamber again. After another 30 seconds the data will have stabilized and is now considered Valid data. These steps are repeated until the Test is stopped. NOTE: The initial inspiratory O2 and CO2 concentrations should be entered in the Subject Data window. These values will be used for the initial 10 minutes of the Test. These initial values will also be applied for the entire test if the Sample Inspiratory Air checkbox is un-selected. NOTE: This feature is recommended only for studies of 30 minutes or more AND in an environment in which the ambient air is likely to change during the course of the test [for example: a small room with several people working near the subject]. The Expiratory Flow Measurement checkbox should NOT be checked. To allow for measurement of Flow on the expiratory side contact AEI Technologies for assistance. Cal Bypass checkbox should NOT be checked. To allow bypassing the Interface Box for gas calibration contact AEI Technologies for assistance. The Quick Cal checkbox should NOT be checked. To allow for a shortened gas calibration time contact AEI Technologies for assistance. Click ‘OK’. The software will automatically re-initialize. Completely close the program. Re-start the program. 7 Setup Defaults IMPORTANT! Required for proper operation • In the Menu Select: File > Properties > Setup tab: • Change ‘Standard Calibration Gases’ values to your Laboratory Calibration Gas values. Enter the value in the appropriate box. • Enter the approximation of ambient inspired gas values (to be measured and adjusted later, after calibration of the analyzers). FiO2 = 20.93% typical for fresh [outdoor] air; FiCO2 = 0.04% typical for fresh [outdoor] air. • Confirm that the Mixed Volume Delay is set at 6.9 Liters. This is for the supplied breathing valve and tubing (2.7 liters) and the mixing chamber (4.2 liters). If different components are used refer to Appendix A. • The default Analyzer Time Delays are 3 Seconds for the CO2 Analyzer and 3.3 seconds for the O2 Analyzer. NOTE: Systems variations may alter these delay times. For most accurate phasing refer to Appendix A. • Confirm that the Calibration Syringe volume is set to the supplied syringe volume as recorded on the provided syringe calibration certificate, or enter your own Calibration Syringe volume. • Confirm that the Volume correction [Ve and Vt only] is calculated in BTPS (preferred) or STPD. NOTE: All metabolic data [VO2, VCO2, etc.] is always presented and saved in STPD without regard to this selection box. 8 Averaging Interval • • In the Menu Select: File > Properties > Avg Interval Choose the Averaging Interval for the test by sliding the bar to the desired setting. NOTE: By setting the Averaging Interval to 0 seconds you will get breath-by-breath data. However, the exhaled breaths are still sampled from the mixing chamber. Institution Name & Address In the Menu Select: View > Preferences. Fill in the Institution Name and Address. This information will appear at the top of the printed reports. 9 DAILY SYSTEM STARTUP & SHUTDOWN Each day, before System Calibration or starting a test, perform the following: STARTUP • Turn the Range Knob on the right of the S-3A/I O2 Analyzer to the T.C. Setting. The value displayed on the panel meter should in be 6.790 (+/- 0.003). If the displayed value falls outside of this range, wait and allow time for the value to stabilize. • Turn ON the R-1 Pump. Adjust the black, needle valve knob below the flow meter until the bottom of the lower SILVER float ball is set at the 2 on the graduated meter. This represents a gas sample flow rate of approximately 175 ml/min. Refer to Analyzer manuals for flow rate graphs. NOTE: DO NOT CHANGE THIS SETTING DURING AND AFTER SYSTEM CALIBRATION IS PERFORMED. Changing the sample flow rate will adversely alter calibration. • Turn ON the Interface Box, Computer and Printer. It is also recommended that the S-3A O2 Analyzer and CD-3A CO2 Analyzer always be left powered ON. This will prolong the life of the analyzers and greatly decrease the system warm-up time. • Start the AEI Metabolic System Software program by double clicking the icon on your desktop. A box of text in the lower right corner of the screen asks you to ‘Please Wait’. Wait until this box disappears. When the software has been loaded properly this box will disappear and the software is ready for use. NOTE: Do not attempt to operate the software until the ‘Please Wait’ message disappears. SYSTEM LEAK CHECK • Turn the R-1 Pump Power ON. • Temporarily disconnect the small-diameter sample tube from the Mixing Chamber with a quarter-turn of the Leur connector. • Using your finger [or a bend the tubing] to close off the tube opening. • Verify that both balls on the flow meter drop completely to the bottom of the tube and stay there with no bouncing (Zero flow). • Remove your finger and verify that the SILVER float returns to a setting of 2 on the Flow meter and that the Analyzer readings return to 20.93 % and 0.04%. • Re-connect the sampling tube to the Mixing Chamber with a snug fit. • Wait at least 10 minutes before System Calibration. CALIBRATION GAS TANKS • Fully open the Calibration Gas tank valves [>1 full turn counter-clockwise] and adjust both Calibration Gas regulators to 15 psi. DRYER DESICCANT • Observe the small desiccant beads through the window of the Dryer Box. The beads should be a deep blue color. When any of the beads start to change color from blue to either purple or pink then it is time to change the desiccant. To change the desiccant slide the cover [with an arrow and ridges] off by pressing down on the inner edge of the cover and at the same time sliding it out. Discard all the desiccant and add new desiccant such that the beads completely fill all empty space in the box. Replace the cover. 10 At the end of the day: CALIBRATION GAS TANKS • Turn off the Calibration Gas for both cylinders. Use the cylinder wrench to shut off the gas supply by securely tightening the main cylinder valve in a clockwise direction. NOTE: Do not over tighten. S-3A OXYGEN ANALYZER CELL RESTORE • Once every week press the Cell Restore on the front panel of the S-3A/I O2 Analyzer. This should typically be done at the end of the day’s testing because the analyzer takes about 1 hour to recover from this procedure. o Turn the R-1 Pump Power. o Wait about 15 minutes until the Cell Restore light on the front panel extinguishes. o Turn Power OFF on the R-1 Pump POWER OFF • It is recommended to turn Power OFF on the R-1 Pump, Interface Box, computer, and Printer at the end of each day’s testing. It is also recommended that the S-3A O2 Analyzer and CD-3A CO2 Analyzer always be left powered ON. This will prolong the life of the analyzers and greatly decrease the system warm-up time. 11 SYSTEM CALIBRATION Gas Calibration and Verification and Volume Calibration and Verification should be performed before every test. When used for teaching, demonstration, and testing where data will not be published, Gas Verification and Volume Verification may be sufficient; and then re-calibrate only when necessary. IMPORTANT NOTE: Always perform Daily System Startup before System Calibration and Validation. System Calibration window Gas Calibration: O2 and CO2 NOTE: For best results both S-3A O2 analyzer and CD-3A CO2 analyzer must be Powered On for 16 hours minimum before calibration. • Click on the ‘Calibration’ button on the upper left of the screen. • In the Valve Control section on the right side of the System Calibration window select ‘Calibrate Exp’. This will select the tank with typical expiratory gas values; for example 12 16%O2 and 4%CO2. • Wait 1 minute for the gas analyzers to stabilize. • Calibrate the S-3A O2 analyzer: o Turn the Range Switch to the %O2 position. o Turn the CALIBRATE/SAMPLE SWITCH to the SAMPLE position. o Slide the black lever on the ZERO POT to unlock the dial. o Set the ZERO POT to mid-range (“5” on the vernier counter). o Turn the CALIBRATE/SAMPLE SWITCH to the CALIBRATE position. o Adjust the ZERO POT to obtain the exact O2 calibration gas value on the S-3A analyzer panel meter. NOTE: If you run out of range on the ZERO POT: set the CALIBRATE/SAMPLE SWITCH in the SAMPLE position; turn the ZERO POT all the way back in the opposite direction; set the CALIBRATE/SAMPLE SWITCH in the CALIBRATE position; adjust the ZERO POT. Repeat until the panel meter reads correctly. o Turn the CALIBRATE/SAMPLE SWITCH back to the SAMPLE position. o Slide the black lever on the ZERO POT to lock the dial. • Calibrate the CD-3A CO2 analyzer: o Turn the CALIBRATE/SAMPLE SWITCH to the SAMPLE position. o Slide the black lever on the SPAN POT to unlock the dial. o Set the SPAN POT to mid-range (“5” on the vernier counter). o Turn the CALIBRATE/SAMPLE SWITCH to the CALIBRATE position. o Adjust the SPAN POT to obtain the exact CO2 calibration gas value on the CD-3A analyzer panel meter. NOTE: If you run out of range on the SPAN POT: set the CALIBRATE/SAMPLE SWITCH in the SAMPLE position; turn the SPAN POT all the way back in the opposite direction; set the CALIBRATE/SAMPLE SWITCH in the CALIBRATE position; adjust the SPAN POT. Repeat until the panel meter reads correctly. o Turn the CALIBRATE/SAMPLE SWITCH back to the SAMPLE position. o Slide the black lever on the SPAN POT to lock the dial. • In the Valve Control section on the right side of the System Calibration window select ‘Calibrate Air’. This will select the tank with typical ambient air gas values; for example 20.94%O2 and .03%CO2. • Wait 1 minute for the gas analyzers to stabilize. • Calibrate the S-3A O2 analyzer: o Turn the Range Switch to the %O2 position. o Turn the CALIBRATE/SAMPLE SWITCH to the SAMPLE position. o Verify that the S-3A analyzer panel meter value is within +/- 0.02% O2 of the O2 calibration gas value. o If it does not verify, repeat the O2 calibration using the ‘Calibrate Exp’ calibration gas. If still not correct please contact AEI Technologies service. o Turn the CALIBRATE/SAMPLE SWITCH back to the SAMPLE position. • Calibrate the CD-3A CO2 analyzer: o Turn the CALIBRATE/SAMPLE SWITCH to the SAMPLE position. o Slide the black lever on the ZERO POT to unlock the dial. o Set the ZERO POT to mid-range (“5” on the vernier counter). 13 • • • • • • • • • • • • • o Turn the CALIBRATE/SAMPLE SWITCH to the CALIBRATE position. o Adjust the ZERO POT to obtain the exact calibration gas value on the CD-3A analyzer panel meter. NOTE: If you run out of range on the ZERO POT: set the CALIBRATE/SAMPLE SWITCH in the SAMPLE position; turn the ZERO POT all the way back in the opposite direction; set the CALIBRATE/SAMPLE SWITCH in the CALIBRATE position; adjust the ZERO POT. Repeat until the panel meter reads correctly. o Turn the CALIBRATE/SAMPLE SWITCH back to the SAMPLE position. o Slide the black lever on the ZERO POT to lock the dial. In the Valve Control section select ‘Standby’ to stop the flow of gas. Verify Low Cal Value and High Cal Value on the System Calibration window exactly equal the Calibration Gas values of the tanks. If needed, enter new values as follows: o Place cursor and click in the Low Cal Value Box for the Oxygen Channel and enter the exact value of the lower O2 Calibration Gas that you will be using (usually 15 – 16% O2). o Place cursor in the High Cal Value Box for the Oxygen Channel enter the exact higher O2 Calibration Gas value to be used (preferably 20 – 21% O2). o Perform the same steps for the CO2 Channel. Enter the exact higher CO2 Calibration Gas that you will be using (preferably 4 - 6% CO2) and the exact lower CO2 Calibration Gas that you will be using (usually 0 – 0.03% CO2). o Click “Save” Click the channel select box for the O2 Channel (Top Left side of the window) Click on the LO CAL button on the System Calibration window. Wait about 90 seconds until the cycle is complete and the O2 Channel is “deselected’. Select the O2 Channel again. Click on the HI CAL button on the System Calibration window. Wait about 90 seconds until the cycle is complete and the Status box at the right of the O2 Channel will then read “Calibrated”. If the channel indicates “Error” press the Save button, if available, and repeat the software calibration steps. Several repetitions may be necessary. Click the select box for the CO2 Channel. Click on the LO CAL button on the System Calibration window. Wait about 90 seconds until the cycle is complete and the CO2 Channel is “deselected’. Select the CO2 Channel again. Click on the HI CAL button on the System Calibration window. Wait about 90 seconds until the cycle is complete and the CO2 Channel Status will read “Calibrated”. If the channel indicates “Error” press the Save button, if available, and repeat the software calibration steps. Several repetitions may be necessary. Click “Save” Gas Verification NOTE: For best results both S-3A O2 analyzer and CD-3A CO2 analyzer must be Powered On for 16 hours minimum before calibration. 14 • • • • • • • • • • • • • • Click on the ‘Calibration’ button on the upper left of the screen. In the Valve Control section of the System Calibration window select ‘Calibrate Air’ Wait 1 minute for the analyzers to stabilize. Verify that the MixO2 Average Data is within +/- 0.01% of the High Cal Value. Also verify that the MixO2 Average Data is within +/- 0.02% of the values displayed on the S-3A analyzer panel meter. NOTE: the MixO2 Average Data value is used in the metabolic calculations – not the analyzer panel meter value. Verify that the MixCO2 Average Data is within 0.01% of the Low Cal Value. Also verify that the MixCO2 Average Data is within +/- 0.02% of the values displayed on the CD-3A analyzer panel meter. NOTE: the MixCO2 Average Data value is used in the metabolic calculations – not the analyzer panel meter value. If the values are not within acceptable tolerances then perform the Gas Calibration and Verification again. In the Valve Control section of the System Calibration window select ‘Calibrate Exp’ Wait about 1 minute for the analyzers to stabilize. Verify that the MixO2 Average Data is within +/- 0.01% of the Low Cal Value. Also verify that the MixO2 Average Data is within +/- 0.02% of the values displayed on the S-3A analyzer panel meter. NOTE: the MixO2 Average Data value is used in the metabolic calculations – not the analyzer panel meter value. Verify that the MixCO2 Average Data is within 0.01% of the High Cal Value. Also verify that the MixCO2 Average Data is within +/- 0.02% of the values displayed on the CD-3A analyzer panel meter. NOTE: the MixCO2 Average Data value is used in the metabolic calculations – not the analyzer panel meter value. If the values are not within acceptable tolerances then perform the Gas Calibration and Verification again. In the Valve Control section of the System Calibration window select ‘Sample Air’ Verify the MixO2 Average Data indicates a stable reading in the range 20.50 – 20.96 %O2. NOTE: Factors such as smog and altitude may effect the O2 concentration. Verify the MixCO2 Average Data indicates a stable reading in the range 0.01 – 0.15 %CO2. NOTE: Factors such as smog and altitude may effect the CO2 concentration. Volume Calibration NOTE: For Canopy System Volume Calibration see Appendix F. • • • Prepare for Volume Calibration by removing the mouthpiece or mask from the nonrebreathing valve. Make certain that the saliva trap and black cap are screwed on tightly to the breathing valve. Make certain that the inspiratory and expiratory ports of the non-rebreathing valve are also screwed in tightly. The corrugated breathing hose should be snug on the expiratory port. The Pneumotach, if used, should be inserted snugly into the inspiratory port of the non-rebreathing valve; or, alternatively, a breathing hose should be snug on the inspiratory port and Pneumotach [refer to Installation Instructions]. [The black end of the turbine cartridge, if used, should be inserted snugly into the inspiratory port of the breathing valve.] 15 • • • • • • • • • Attach the 3-Liter Calibration Syringe directly to the mouthport connector on the nonrebreathing valve where the mouthpiece is normally attached. Be certain the syringe plunger is pushed against the stop. NOTE: The syringe that is supplied is calibrated to National Standards [NIST], and must not be adjusted without being recalibrated to a known standard volume.) Do not operate the syringe until after opening the Volume Calibration or Volume Verification window. On the right hand side of the System Calibration window you will find a small box labeled “Pneumotach” [or "Turbine"]. In this box, select ‘Calibration’. A Volume Calibration window will be opened. Stroke the Calibration Syringe. While stroking the syringe, the user must apply a full stroke each time, stopping against the mechanical limits of the syringe without banging against the stop. For exercise testing, a stroke rate of 4 seconds [45 L/min] for each outward stroke is recommended for best results; for resting energy [canopy], a stroke rate of 7-8 seconds [24 L/min] for each outward stroke is recommended. Use of a clock second-hand or metronome produces better consistency. After the first two strokes, the relative volume of each stroke measured by the MOXUS System is displayed. After 5 or 6 complete strokes, look at the displayed Relative Vol. NOTE: The values may not represent an accurate volume [in fact, it may be totally erroneous] at this time, but the displayed volumes should be consistent. If the volumes are not consistent click ‘Recalibrate’ to repeat Volume Calibration. The inconsistency may be due to: the strokes may not have been full strokes, or there may be a leak in a tube or a leak (or backflow) in a component of the nonrebreathing valve. Select ‘OK’, and the Volume Verification window will appear. 16 Volume Verification • • • • • • • • Stroke the Calibration Syringe. While stroking the syringe, the user must apply a full stroke each time, stopping against the mechanical limits of the syringe without banging against the stop. For exercise testing, 5 strokes at a stroke rate of 1 to 5 seconds [36-180 L/min] for each outward stroke is recommended; for resting energy [canopy], 5 strokes at a stroke rate of 6-8 seconds [22-30 L/min] for each outward stroke is recommended. Each stroke of the pump will be recorded in milliliters on the Volume Verification Window and the actual error and percent error for each stroke will also appear. After the first two strokes, the Volume of each stroke measured by the MOXUS System is displayed. After 5 or 6 complete strokes verify that the Avg Error is within +/- 2%. Typically, +/1% or better can be achieved. If so, simply Click OK and the window will close. Alternatively, the ‘Clear Grid’ button may be pressed and a different stroke speed may be verified. The ‘Clear Grid’ button will clear the grid and establish the zero flow condition for the Pneumotach. If the error is greater than the tolerance of +/- 2%, you may click on the Recalibrate button to re-calibrate the flow. The Volume Calibration window will open and you will be prompted to pump the calibration syringe at least another 5 times. Once the re-calibration is complete, you conduct another verification to confirm that system is now measuring the volume within +/- 2%. Repeat this process until you complete an effective calibration and verify that the system is operating within the acceptable tolerance range. Click ‘Save’ Calibrating Other Channels This must be done by providing an analog input, which is equal to a known “low” calibration value, and another input, that is equal to a known “high” calibration value. You must enter the high and low Cal values, and name the channel. Calibration of the MOXUS System is now complete and you are ready start a test. 17 CONDUCTING A TEST Gas Calibration and Verification and Volume Calibration and Verification should be performed before every test. When used for teaching, demonstration, and testing where data will not be published, Gas Verification and Volume Verification may be sufficient; and then re-calibrate only when necessary. IMPORTANT NOTE: Always perform Daily System Startup before System Calibration and Validation. Subject Information • • • • • • • • Turn off Canopy Pump, if used. Click on the ‘Subject Data’ hot button on the Toolbar. If a previous test is currently open [i.e. the data is displayed on the screen] then a message will appear asking if you would like to clear the existing subject data: o Click ‘No’ if you would like to retain the existing Subject Data for the next test; or if you are not ready to start the next test. o Click ‘Yes’ if you would like to start a new test and re-enter the Subject Data for the next subject. Enter the Subject Name. NOTE: Do not use special characters such as “/”, “.”, “, etc. The program will add the date and time to the Subject Name to create the File Name and automatically save the file for this test under this File Name in the MOXUS Metabolic System subdirectory [typically: c:\Program Files\AEI Technologies\MOXUS]. Enter the height, weight, age, and sex for the test subject. NOTE: Units conversion is automatic. Enter the room [ambient] barometric pressure, temperature and relative humidity. NOTE: Units conversion is automatic. Measure and enter ambient [inspiratory] fiO2 and fiCO2 concentrations: o Select Menu: Tools > Calibration to open the System Calibration window. o Click on the Sample Air button on the System Calibration window. [NOTE: For Interface Box delivered before January 2014 temporarily disconnect the small-diameter sample tube from the Mixing Chamber with a quarter-turn of the Leur connector]. o Wait at least 60 seconds to allow the MixO2 and MixCO2 Average Data readings on the System Calibration window to stabilize. o Check that the values are reasonable. Nominal indoor ambient O2 is 20.50 to 20.96 and CO2 is 0.01 to 0.15; however, your typical values may be slightly outside these ranges. Re-calibrate if the values are not typical. o Record the Average Value MixO2 and MixCO2 readings on the Subject Data window for fiO2 and fiCO2 respectively. o Click on the Standby button on the System Calibration window. [NOTE: For Interface Box delivered before January 2014 re-connect the sampling tube to the Mixing Chamber with a snug fit]. o Exit the System Calibration window. You also have the option of entering up to 3 lines of Comments for this particular test run. These comments become a permanent part of the data file and they will appear on the Summary Page of the Exercise Report that you can print out following the 18 • • • test. Optionally, Resting HR, Resting BP, and MVV may be entered. The Test Date and Test Time are automatically entered. Click ‘OK’. Test Preparation • • • • • • • • Reattach the mouthpiece or mask to the breathing valve Prepare the Test Subject: o Get the Test Subject in position (on the treadmill, ergometer, bed, etc.) to begin testing. o If you plan to utilize the headgear to support the breathing valve, fit it onto the subject at this time. Alternatively, if you are planning to utilize the Support Arm on the cart, you should position the arm so that the attached breathing valve and mouthpiece are placed directly in front of the subject. o If you are using the Polar Telemetry System to monitor heart rate, you should moisten the underside of the chest belt and strap it into position on the subject at this time. Please place the Polar Receiver as close as possible to the subject by taping it to the treadmill handrail or ergometer handlebar. o If a mouthpiece or mask is used: Instruct the subject to insert the mouthpiece completely into the mouth and position a nose-clip in place; or attach the mask to the subject ensuring that there are no leaks. o The subject should begin to breathe normally to flush the mixing chamber and fill it with exhaled gases before starting the test. Click on the ‘Start Test’ button on the Toolbar. This opens the Real-Time Data window for a final check of the system operation prior to the actual start of the test and saving of data. Before performing a test using the Canopy Hood wait about 5 seconds, and then turn on the Canopy Pump. Refer to Appendix F. Wait about 30 seconds for the numeric values on the Real-Time Data window to start to change and then stabilize. Check the numeric values on the Real-Time Data window to be certain that the tidal volume and ventilation is being measured and that the FeO2 and FeCO2 values have changed from ambient to reflect the concentrations in the expired breaths. Verify also that these values are tracking the readings on the individual analyzer panel meters. If heart rate is being measured verify that the “heart symbol” is flashing and that the heart rate is being displayed appears correct. If a Treadmill or Ergometer is connected verify that the Work/Grade and Speed are correctly displayed. Start the Test • • Click on the flashing Start Test button at the bottom right of this window. The Real-Time Data window will minimize and 3 new windows will appear: o At the top, 2 Data Graphs will appear to track parameters during the test run. o On the bottom, the Tabular Data window will appear. o The test data shown is based on the Averaging Interval that you selected on 19 • • • the Avg Interval window. o The Real-Time Data window can be displayed by clicking on ‘Window’ in the menu and selecting the Real-Time Data window. Two minutes following the beginning of a test, ventilation, gas analysis and heart rate signals are tested for validity. Should any or all of the signals be found to be obviously unacceptable, a warning prompt is displayed and the operator is given a choice to continue or terminate the test. Data is collected during this message. During a Test you may perform the following as explained in the next section of this Manual: o Data Graph configurations o Add another Graph o Tabular configurations o Averaging Interval changes o Manual entry of off-line data, such as treadmill speed or workload. o Cardiac Output tests (with optional equipment. See Appendix E for instructions on performing cardiac output studies) All data is automatically be saved in the AEI Metabolic System Software subfolder [typically, C;\Program Files\AEI Technologies\Moxus]. End the Test • • Once the test is complete click on the ‘End Test’ button on the Toolbar. o A small window will open asking if you are sure that you want to end the test. o Click YES to stop data collection and end the Test. NOTE: to start another test enter new subject information, even if using the same subject. 20 RUN TIME FEATURES Data Graphs Data Graphs may be changed and rescaled within the Properties window by clicking on the desired Data Graph to select it then right-clicking over the desired graph. Once in the Properties menu, select the “Graphs” tab. NOTE: The Data Graphs should only be changed or added during the first 10 minutes of a Test; otherwise Data Graphs may be changed after the test is complete. • • • • • The axis parameters are then selected for X axis, Primary Y Axis and Secondary Y Axis. If the Secondary Y Axis is not to be used select ‘<none>’. Manual scaling is the default setting. Enter the desired limits in the Lo and Hi boxes by inserting a numeric value. Automatic scaling is not recommended for general use. The graph is scaled automatically, and “zooming” is enabled, if the ‘Auto Scale’ box is selected. Grid markings may be placed on the graph by checking the Grid checkbox. Unclick it to remove grid marks. When comparing two Y parameters, it is often useful to have the scaling equal to each other. The operator can do so by checking the ‘Use Primary Axis’ checkbox. If it is left unchecked, the graph scaling will be set as indicated in the Lo and Hi settings. Additional Data Graphs may be added by selecting in the Menu:Tools>Add Graph. 21 Tabular Display Parameters may be altered in the tabular display by means of right click on the tabular data. When right clicking in the tabular data section, the following menu is displayed. NOTE: The Tabular Display is limited to 9457 rows. Longer Averaging Intervals allow data to be viewed for longer tests. Export to Excel NOTE: It is generally not advisable to export data to Excel during a test; Export to Excel should be performed after the test is complete. • Click on the box marked ‘Export to Excel’. The Export Window will open. • The Excel file is saved in the AEI Metabolic System Software subfolder [typically, C;\Program Files\AEI Technologies\Moxus]. However, you can change this setting by clicking on the folder icon and setting the destination to the folder of your choice or to a CD. • Fill in the File Name that you choose for the Excel file. • Click OPEN. The program will export your data file as a new .xls file to the destination that you have selected. • The newly created Excel file is now ready to be opened. • There will be two tabs within the Excel file: Subject Data and Tabular Data. • The only initial adjustment that will have to be made within Excel is to expand the column widths so that all the subject data, parameter headings and numeric values fit into the columns properly. • The file can now be manipulated normally as a standard Excel spreadsheet. 22 Edit Data NOTE: It is generally not advisable to Edit Data during a test; Edit Data should be performed after the test is complete. The Edit Data function allows the user to disable (or re-enable) individual breaths that, due to aberrations such as sighs, swallowing, coughing, talking or tubing artifacts, might skew the true average. Generally speaking, longer averaging intervals will lessen the need to disable aberrant breaths. To disable a breath, choose ‘Edit Data’ from the menu that is presented by right clicking on the tabular data. The Tabular Data is then changed from an averaged breath display to a breath-by-breath display. The data for each breath can then be reviewed. Breaths that need to be disabled are chosen by double-clicking on the breath number at the far left of the tabular display. A “disabled breath” is displayed as a row of faded text. Double-click on the “12” in the left hand column to disable the breath at 00:31 Conversely, a disabled breath may be re-enabled by double-clicking on the breath number. The text will be displayed normally. Once editing is complete, right click on the tabular data and select ‘End Data Edit’ to return to an averaged data display. Hide/Unhide Columns The Hide and Unhide functions in the Right Click Tabular Menu allows the user to remove and add back columns from the Tabular Display and Tabular Report. Simply Select the column you wish to remove and then right-click to bring up the menu. Selecting ‘Hide’ removes the column from the table. The hidden columns are permanently saved as hidden columns from the Tabular Display (but not hidden in the Tabular Reports) until unhidden by the ‘Unhide’ function. The Hidden column is indicated by a slightly thicker dividing line in the column heading. To Unhide, Select the column immediately to the right of the column you wish to unhide. Selecting ‘UnHide’ displays the parameter in the table again. Selecting ‘UnHide All’ will display all the columns of data. Column Move From/Move To These functions are complimentary and allow reordering of the data columns. For example, to move RER to the column next to Freq, Select the RER column. Then select ‘Move from’. 23 Next, select the Ve column (the column where you want to place RER) and select ‘Move to’. Ve will be pushed to the right and RER will be placed in its column. The moved columns are permanently saved in the Tabular Display (but not changed in the Tabular Reports). If needed, to restore the columns to their Default order: close and then open the program; open any saved data file; in the Tabular Display right-click and select ‘UnhideAll’; then select ‘UnHide’; then select ‘MoveFrom’; last select ‘MoveTo’; close the program. The columns are now restored to default locations. Sort Column Ascending / Descending Allows reordering of data depending upon the column that is selected when choosing these functions. This is most useful when you wish to display the end of the test at the beginning of the column. To do so, Select the Time column. Then right click and select ‘Sort Descending’. The Time column can be returned to the original state by selecting it and then selecting ‘Sort Ascending’. Select Column Selects a column for use with other menu functions such as Sort Ascending, Move etc. A column can also be selected by clicking the mouse on the desired column header. Properties Displays the properties menu for selecting choices for graphs, averaging intervals, setup parameters, etc. 24 Mark Invalid/Valid Data Allows elimination of selected data rows that are printed on the report. This function is most useful to eliminate the warm up and cool down periods of a test thereby reporting only on the exercise protocol of the test. Select the data row(s) that you wish to be removed from the report. Next, select “Mark Invalid Data” from the Tabular Right-Click menu. To mark the data rows as Valid again: select the data row(s) that you wish to be added back to the report. Next, select “Mark Valid Data” from the Tabular Right-Click menu. NOTE: the data cannot be marked a valid while performing a test. Column Width The width of each column may be adjusted by placing the mouse over the column line in the heading such that a ‘double arrow’ appears. Hold down the left mouse button and adjust the column width as desired; then release the mouse button. The column width is permanently saved in the Tabular Display (but not changed in the Tabular Reports) Averaging Interval Change the Averaging Interval by placing the pointer on the graphs and right-clicking the mouse. The Properties window will appear. Move the sliding bar to adjust the interval. NOTE: Data from every breath is always collected and saved by the software independent of the Averaging Interval selected. The Averaging Interval selection is for display purposes only. Setting the Averaging Interval to ‘0’ will display Breath-By-Breath data. Changing the Averaging Interval to ‘0’ should only occur during the first 10 minutes of a Test; otherwise it should be changed after the test is complete. 25 Data Entry The Data Entry button allows the user to manually enter certain numeric data at any desired time during a test. This is useful, for example, for manual entry of Heart Rate, Speed, etc; or using USER to mark data. Cardiac Output tests (with optional equipment) See Appendix E for instructions on performing cardiac output tests. 26 OTHER FEATURES Full Screen Graphs/Chart View the Full Screen display of Data Graphs or Tabular Data by clicking on the “maximize” window square in the upper right corner of the desired window. Click on the “Restore” window square to replace the window to its original position. ZOOM NOTE: The ZOOM feature is enabled only when “Auto Scale” is enabled. • The Graph function also includes a ZOOM feature. • As an example, if the total test includes 10 minutes of data, each graph that you callup will display the parameters you choose over 0-10:00 of the test. • If you would like to see a graph displaying only those data points collected during the 3:00 to 5:00 time frame, you can set the graph to zoom into that specific period. • This can be done within a particular graph by placing the pointer inside the graph on the starting time that you want to zoom into (the 3:00 mark of the test) and leftclicking the mouse. • Hold the left-click and move the pointer to the end time within the graph that you want to zoom into (for example the 5:00 mark of the test). • The Graph will now re-set and zoom-in to display only the data collected between the 3:00 and 5:00 marks of the test. • Restore the full-scale graph by selecting File > Properties or simply by right-clicking on the graph to bring up the Properties window. Click on the ‘Graphs’ Tab. Click the ‘OK’ button to restore the full-scale graph. File Open • In the Menu Select: > File > Open and a window will open which will list all of the data files (.dat) in the MOXUS Metabolic System folder. • Open a file by double-clicking on the name of the specific data file that you want to view. • When the file is opened, the full screen display will include two graphs in the top half of the screen and the tabular data record of the test in the bottom half of the screen. Real-Time Graph The Real-Time Graph feature is enabled by selecting the ‘Real-Time Graph Enable’ checkbox on the Real-Time Data window. Each Real-Time Graph in the Real-Time Data window is enabled or disabled in the Graph Selection window by clicking on the box to the left of each parameter. To change the scaling, click on the desired parameter in the Graph Legend window, enter the desired minimum and maximum scale values in the boxes to the right and click ‘Apply’. 27 28 REPORTS Print Reports Exercise Summary Report NOTE: Not available for Canopy System, see Appendix F. • Click on REPORTS > EXERCISE to Print the Report. • The Report includes the full tabular data chart for the test as well as a Summary Page with Resting / Max Achieved values and comparisons to predicted Max values for the subject. Energy Expenditure Report • Click on REPORTS > ENERGY to Print the Report. Calibration Report • Click on REPORTS > CALIBRATION to Print the Report. Print Preview To preview the reports before printing select FLIE>PREVIEW>[Report] Setup Graphical Reports Go to “FILE” and select ‘OPEN’. Select a file with data in it. Press ‘OPEN’, and go to “REPORTS” and select ‘SET-UP’. Select the display of the page that you want to change first. IN THE EXAMPLE ABOVE, PAGE 1 IS SELECTED Scroll through the page to display the image of the graph to be changed, and left click on that graph. Select the tab “DEFINITION”. Select the desired “x”, “y1”, “y2” data to be 29 displayed on that graph. If you wish the axis for the two “y” axis to be the same, select ‘USE PRIMARY AXIS’. Select each of the graphs that you wish to change, sequentially, and select each page in turn. SETTING CONFIGURATION OF GRAPH TO LEFT BY LEFT CLICKING GRAPH NOTE: THE “X” AXIS IS ALWAYS THE HORIZONTAL AXIS, AND THE “PRIMARY Y AXIS” AND “SECONDARY Y AXIS” ARE ALWAYS THE LEFT AND RIGHT AXIS, RESPECTIVELY. ADDING ADDITIONAL PAGES OR GRAPHS Select ‘SET-UP’, and select a page. Select the ‘PAGE CONFIGURATION’ tab, then select ‘INSERT BEFORE PAGE’ or ‘INSERT AFTER PAGE’, and select the desired layout. Left click, with the curser, on the desired graph, or click on ‘GRAPH DEFINITION’ tab. Select the desired data to be graphed. Select ‘SAVE’. A “SAVE REPORT” dialog box will open. When you select ‘SAVE’, a prompt will appears warning that “GRAPH.RPT” already exists. Click ‘YES’ to save the changes. Otherwise, click ‘NO’ to discard changes. 30 APPENDIX A THEORY OF OPERATION & FORMULAS System Modes The method the AEI Metabolic System Software collects, displays, saves and reports data is dependent upon the testing methodology and system configuration selected on the Hardware Diagnostics window: 1. When utilizing a mouthpiece or mask with a human subject [MOXUS or MAX-II System mode], and sampling gas from a mixing chamber, data is acquired on a breathby-breath [i.e. 1 data point per breath] basis. 2. When sampling from an animal chamber [MOXAR System mode] data is acquired on a 1 data point per second basis. 3. When utilizing the system for general Data Acquisition [DATA ACQ System mode] data is acquired on a 1 data point per second basis. 4. When utilizing the Canopy hood on a human subject [Canopy System Option] data is acquired on a 1 data point per second basis. 5. When a test will be collecting data for over 6 hours Long Term Monitoring is required [Long Term Monitoring option]. Averaging Intervals of 5 seconds or more should be used. Data is acquired on a 1 data point per second basis multiplied by the Averaging Interval. NOTE: Changes to display, table, or operational parameters during a test in Long Term Monitoring mode may result in lost data. If parameters must be changed do so in the first minute. Design Philosophy The AEI Metabolic Systems Software, or MAX-II Software, is the result of a demand for the accurate measurement of oxygen uptake and other parameters during exercise testing or at rest, while allowing maximum flexibility in the system configuration, graphic display and reporting of data. The Software allows the user to change the manner in which he or she views the data while the test is proceeding. In addition, the Software provides the ability to set up multiple configurations to minimize setup time when switching equipment for different testing methodologies. The AEI Metabolic Systems Software is utilized in a variety of AEI Technologies systems and product options. These include: The MOXUS, MAX-II, and MOXAR Metabolic Systems; Canopy System and Cardiac Output options; and Long Term Monitoring and DAQ modes. NOTE: all of these selections may not apply to the system you purchased. In order to achieve maximum accuracy, the Software uses a breath as the basic unit upon which all data is calculated [MOXUS and MAX-II System modes only]. As the program corrects all phase delays between the gas analyzers and the other “real time” data, the data displayed for each breath is precisely corrected. Facilities are included to change the delay factors to allow for changes in sampling rate, tubing length, and other alterations to the apparatus. We utilize an active mixing chamber for the exhaled gas, thereby smoothing out rapid breath-by-breath variations and producing more accurate data for most applications. 31 During exercise testing, spurious data may be sent to the computer. This may be caused by a loose heart rate sensor, the mouthpiece or tubing falling off, the subject swallowing and possible other small occurrences. This Software does not allow the user to erase or edit any data, however, the erroneous data, which is collected during one of these episodes, may be “Marked Invalid” (ignored). The data will be retained in the user’s data file, but will be excluded from averaging in the final report and on the graphs that are printed by the software. Such Marked Invalid data may, if desired, be reselected as Valid again. Users may also select the time interval that is of interest to them during any test and plot or print only that data, ignoring portions of the data [by marking Invalid] falling outside of the selected time period. For instance, the exercise recovery period may be ignored. This Software allows the operator to enter certain data, manually, during a test, and may be labeled by user via Manual Data Entry. This data may be printed against other data. This feature allows data to be stored and compared to other data. Analog to Digital Conversion Analog to Digital conversion of all analog input signals occurs at 250 Hz with 16 bits of resolution [3rd generation only]. Each channel has an averaging value that is preset for the frequency content of the data on that channel. The resulting averaged value is transmitted to the AEI Metabolic System Software computer, via the USB cable, as a single data point: either on a breath-by-breath basis or a 1 data point per second basis. Saved Data All data points sent to the AEI Metabolic System Software are continuously saved every 30 seconds in a file with a .dat extension and a prefix containing the Subject Name, Test Date and Test Time. For example: If you entered - John Smith – as the name on the Subject Data window, and the subject data was entered on the 15th of November 2002 at 11:30 [24 hour clock basis], the file will be named John Smith1115021130.dat and it will automatically be saved in the AEI Metabolic System Software subfolder [typically, C;\Program Files\AEI Technologies\Moxus]. This Saved Data file contains “raw data” and it is saved before any calculations are performed on it. However, the MixedO2 and MixedCO2 channels, breath-by-breath sampling only, are delayed in time before saving. The Saved Data file is viewable by the user only when opened using the AEI Metabolic System Software. Breath-by-Breath Measurements The MOXUS is breath-by-breath metabolic system sampling from the mixing chamber. The user can adjust the phasing delays as appropriate. The Time displayed on the tabular display is the time for a particular breath and it may not exactly correspond with ‘real time’ as displayed in the Time box at the top. This is because there are sometimes delays in communication between the computer and the Interface Box. Breath-by-Breath Phasing Delays Breath-by-breath exhaled gas concentrations are measured and saved after incorporating an appropriate phasing delay. This delay is necessary because of two 32 factors: 1) the time necessary for the expired air to transverse the volume of the nonrebreathing valve, tubing and mixing chamber [Mixed Volume Delay]; and 2) the gas analyzer tubing and response delays [O2 Analyzer Delay and CO2 Analyzer Delay]. Following the end of each breath the system waits until the subject has exhaled a volume at least equal to the value set for the Mixed Volume Delay. It then waits a specified number of seconds for each of the analyzer time delays. It then phases the MixedO2 and MixedCO2 data with the breath that produced that data. It is therefore important to understand how the data is displayed in the graphs, table, and reports during a test. For typical exercise applications where averaged data, and not breath-by-breath data, is desired these delays need not be exact. The software default values for these delays are typical system approximations and are acceptable for most applications. For breathby-breath applications, low tidal volume tests [e.g. resting or child applications],nonstandard system configurations, or critical metabolic measurements it is best to determine the correct time delays. The volume of the non-rebreathing valve, tubing and mixing chamber that is entered for the Mixed Volume Delay can be computed in 1 of 3 ways: 1. measure the actual volume of the components with water and a graduated cylinder. 2. calculate the total volume used from known inside dimensions. 3. a combination of 1 and 2 above. The delay times for the O2 Analyzer Delay and CO2 Analyzer Delay can be computed in 1 of 2 ways. NOTE: these time delays will change proportionally to the sample flow rate: 1. by measuring the actual analyzer response times for a gas to travel from the mixing chamber outlet to each analyzer. Disconnect the sample tubing from the mixing chamber and allow to stabilize. Then quickly apply the calibration gas [remembering to relieve the pressure] and measure each analyzer’s response time to reach 90% of reading. The time can be measured with a stopwatch or using the Real Time Graphs. 2. For each analyzer: calculate the total volume of the sample tubing from the mixing chamber outlet to each analyzer using from tubing inside diameter and tubing length. Then multiply by the sample flow rate to determine the time. Next, add the analyzer’s response time. Ambient Temperature, Pressure and Humidity Ambient [inspiratory air] Temperature, Pressure and Humidity must be entered on the Subject Data window before each test so that an accurate calculation of ventilation can be made. These parameters are used to convert the ATP flow data to BTPS and/or STPD units. Accuracy for these measurements should be: Temperature +/- 1 deg. C; Pressure +/- 2 mmHg; Humidity +/- 5% rh. BTPS and STPD All metabolic data [VO2, VCO2, etc.] is always presented and saved in STPD without regard to the selection box in the Properties/Setup window. The display of Ve and Vt may be displayed in either the default BTPS or, if selected, STPD. 33 The Testing Environment It is strongly recommended that all metabolic data be performed in a room where there is good circulation of outside [fresh] air. Most indoor HVAC [heating, ventilation, airconditioning] systems are adequate for this purpose. In cases where there is no HVAC system of the HVAC system is poor it is best to move the testing to a room with better circulation. If this is not practical then a combination of fans and open windows may be necessary. The ambient temperature and humidity should be reasonable so as not to cause the subject any unnecessary discomfort. The number of persons in the room should be minimized to prevent buildup of CO2 during the test and to minimize disturbing the subject. Flow Measurement The flow signal from a Pneumotach, if used, is sampled and converted to a linearized flow signal using a calibration look up table. This adjustment corrects non-linearity in the Pneumotach, transducer, and electronics. The respiratory phase is determined through the use of a comparator circuit, triggered by flow, which generates a true signal during inspiration and a false signal during exhalation or pause. The integrator is zeroed during the expiratory phase and then accumulates all linearized flow rates during the inspiratory phase. This accumulation ends with the onset of exhalation. A gain factor is then used to convert the accumulated value to an actual volume in liters (inspiratory, ATPS). Warm Up For best results it is strongly recommended that both S-3A and CD-3A gas analyzers be powered on for at least 16 hours before calibration and testing. Also, it is strongly recommended that the R-1 Pump be powered on for at least 10 minutes before calibration and testing. An Uninterruptible Power Supply may be utilized for maintaining a power-on condition if the Moxus must be unplugged to move it to a different location. If necessary, reasonable results [that may not meet specifications] for calibration and testing may be achieved with warm up times shortened to 4 hours for both gas analyzers and 1 minute for the R-1 Pump. Both S-3A and CD-3A gas analyzers should always remain powered on to avoid the warm up period; this will not adversely affect the useful life of the analyzers. Sample Flow The new Interface Box [square plastic box] electronics has internal valves and pressure restrictors to allow for easier calibration. However, this requires that the R-1 Pump flow be set with the bottom ball at 2 [about 175 cc/min] and a regulator pressure of 15 psi and a 1/16”ID sample tubing length of 20” – 24” on the Mixed Inspired port. [NOTE: For Interface Box delivered before January 2014 also connect a 20” – 24” tube to the Air In port]. If a different sample flow rate or tubing length is desired; or if the calibration gas can no longer be connected directly to the Interface Box - please contact AEI Technologies for assistance. The gas analyzers, particularly the S-3A, are sensitive to the sample flow rate. For this reason the sample flow rate must not be changed after hardware calibration of the 34 analyzers. The R-1 Pump will maintain a stable sample flow rate during calibration and testing. Channel Calibration The AEI Metabolic System Software performs a linear calibration for each channel and saves 2 resulting calibration values: Offset and Factor. During a Test each channel data is multiplied by the Factor and then added to the Offset. Offset and Factor values for each channel may be observed on the Calibration Report. Calibration Gas There are a variety of grades of bottled calibration gases available on the market today. We strongly recommend that only Primary Laboratory Standard gases, certified gravimetrically, be used to calibrate the AEI Metabolic Systems. This grade of gas is normally delivered either with a very specific label or an attached certificate stating its contents out to two decimal places. Specified accuracy should be +/- 0.02% absolute or better for concentrations over 2%, +/- 10% of component for lower concentrations. Naturally, utilization of the most precise calibration gases assures you of the most accurate calibration possible for your AEI Metabolic Systems gas analyzers. It is strongly recommended that 2 calibration gas cylinders be used for gas analyzer calibration: typically 21.00%O2 and 0.03% CO2 for one cylinder; and typically 16.00%O2 and 4.00% CO2 for the other. This assures accurate measurement of expiratory gas and for accurately measuring inspiratory [ambient] gas concentration values. If standard outside The AEI Technologies Calibration Gas Module is designed to meet these stringent requirements. Room air is a suitable and convenient choice of calibration gas for some non-critical applications. It must be dried, free of contaminants, and well ventilated with fresh outside air. Heart Rate Detection The heart rate detector is a part of the MOXUS and MAX-II Metabolic Systems. It can accept signals in 2 different forms: the output pulses of a Polar transmitter and receiver; or an amplified analog ECG signal (1 volt per millivolt). The range of the Polar transmitter/ receiver is approximately 2 feet or less, and the best reliability is achieved when the labels on the transmitter and the receiver are positioned in parallel planes. When receiving the analog ECG, the software detects the "R" wave, measures the "R R interval", and computes an average heart rate. RER [Respiratory Exchange Ratio] Variations There are many reasons for observing variations in the measured RER values as compared to the ‘norms’. Some of these reasons include: -RER [the ratio at the mouth] is similar to but different from RQ [the ratio at the muscles]. RER should typically be compared at ‘steady state’ levels of exercise as opposed to changing levels of exercise. -The subject quite often may hyperventilate at the beginning of the test because of unfamiliarity or hurrying; thus raising the RER to 1.0 or higher. 35 -The RER can vary greatly at rest depending upon the substrate utilized, fat or carbs [Wasserman p. 49]. -The RER can vary greatly during the first minute of an exercise test due to retained CO2 [Wasserman p. 43]. Real-Time - These values are measured on a breath-by-breath OR on a 1 sample per second basis during the acquisition of data depending upon the System Mode. They are measured either at the end of the breath or at a later time as determined by a delay factor (see below) during. These measurements form the basic data from which all reported results are generated and are never seen by the user. They are always measured under ambient conditions. This Real Time data is always stored. Breath-by-breath - These values are directly calculated from the real-time results of each breath. As such they may be reported for breath-by-breath results or averaged directly. These represent the conversion of the real time results into user interpretable values. They are appropriately transformed for gas conditions based on the current settings of temperature and pressure and humidity. These values are never stored; rather they are recalculated each time they are needed. Calculated Values - These values are dependent on the breath-by-breath data. They can be calculated either from the breath data or from averaged breath data. These values are not averaged themselves. They are appropriately transformed for gas conditions. Formulas Breath-by-breath Calibration method Real value = (A to D * Factor) + Offset Time of breath Time = time at end of inspiration Ttot Total time of breath Ti Inspiratory time MixO 2 Averaged A to D value of O2 channel with calibration MixCO Averaged A to D value of CO2 channel with calibration 2 Vt - Summation of instantaneous flows with calibration. 36 Haldane Transform and STPD/BTPS correction is applied as required. End Tidal CO Previous Peak value at the time of the breath converted with calibration (Phased for analyzer and sampling delay). 2 Other analog inputs Averaged A to D value converted with calibration Heart rate Averaged Value based on pulse input or precise R-R timing Calculated information from breath by breath or averaged values that may not be directly averaged. Note - in the below formulas “avg” may be either a single breath or the arithmetic average of breaths in a given time period. Frequency F = 60 / Ttot avg avg Minute ventilation Ve = Vt * F avg avg avg Ti/Ttot ratio Ti/Ttot = Ti = Ttot avg avg avg Vt/Ti ratio Vt/Ti = Vt / Ti avg avg Ve/BSA ratio Ve/BSA = Ve / BSA avg Oxygen consumption, Exercise VO = (Vi * FiO ) - (Ve * MixO ); Vi = Ve * (1-FeO -FeCO )/(1-FiO -FiCO ) 2 avg avg 2 avg avg 2 2 avg 2 2 2 CO production, Exercise VCO = (Ve * MixCO ) - (Vi * FiCO ); Vi = Ve * (1-FeO -FeCO )/(1-FiO -FiCO ) 2 2 avg avg 2 avg avg 2 avg 2 Respiratory exchange ratio RER = VCO / VO 2 avg VO2/Ve ratio VO /Ve = VO 2 2 avg 2 avg / Ve VCO2/Ve ratio VCO /Ve = VCO 2 2 avg avg / Ve 2 2 avg 37 2 VO /Kg ratio VO /Kg = VO 2 2 2 avg O pulse O Pulse = VO / Wt(kg) 2 2 2 avg /Heart Rate avg Body Surface Area Body Surface Area (BSA) = .202 x (Wt./2.2) VO /BSA VO /BSA = VO .425 x (Ht./39.37) .725 2 2 2 avg / BSA VCO /BSA VCO /BSA = VCO 2 2 2 avg / BSA THE FOLLOWING VALUES REQUIRE AN END TIDAL CO2 DATA Dead Space VD = (EtCO - CO Mix * P ) / EtCO *Vt -DS Where DS is the valve and mouthpiece dead space 2 avg 2 avg barom 2 avg avg system system VD/VT ratio VD/VT = VD / VT avg avg Alveolar volume VA = Ve - [f *(VD +DS )] Where DS is the valve and mouthpiece dead space avg avg avg system system The following calculations are used for the raw data only and are not used for calculations in the Energy Expenditure Report. Daily urea nitrogen (UN) = Urea Nitrogen / 24 (Note: Corrects patient entry to grams per hour) Protein = (6.25 x 4.2 x UN) Resting Energy Expend. (REE) = (3.94 x VO ) + (1.11 x VCO ) >From Weir Equation 2 2 Adjusted Metabolic Expenditure (AME) = (REE - 2.17) x UN Non protein respiratory quotient (NPRQ) = (VCO - (4.8 x UN))/(VO - (5.9 x UN)) 2 2 Carbohydrate+Fat metabolism (CHOFAT)=AME - Protein % Carbohydrates = (CHOFAT/AME) x ((97.4 x NPRQ) - 68.9)/ ((59.4 x NPRQ) - 30.8) x 100 38 % Fats = (CHOFAT/AME) x ((38.0 x (1.0 - NPRQ))/(59.4 x NPRQ) - 30.8) x 100 The following calculations are used to derive steady state data in the Energy Expenditure Report: Volume of CO produced by protein catabolism (VCO ) = Urea Nitrogen x 4.9 2 2un Volume of O produced by protein catabolism (VO ) = Urea Nitrogen x 5.9 2 2un Nonprotein respiratory quotient (RQ) = (VCO - VCO ) / (VO - VO ) 2 2un 2 2un Kilocalories of energy produced by protein (KCALPRO) = UN x (5.9 x 4.463) Where: 5.9 = grams protein / gram nitrogen 4.463 = Kcal / gram protein Kilocalories of energy produced by fats and carbohydrates (KCALFAT+CHO) = ( 4.686 + RQ - .707 x .361) x ((VO - VO ) .293 2 2un Total Kilocalorie expenditure (KCAL) = KCALPRO + KCALFAT+CHO Total Kilocalorie expenditure normalized to body weight (KCAL / Kg) = KCAL / Body Weight Percentage of total kilocalories from protein (% PRO) = KCALPRO / KCAL x 100 Ratio of Kilocalories of FAT+CHO to Total Kilocalories (KCAL ratio) = KCALFAT+CHO / KCAL Percentage of total Kilocalories from carbohydrates (% CHO) = 504.7 x (RQ - .707) x KCAL ratio 5.047 x (RQ - .707) + 4.686 x (1 - RQ) Percentage of total Kilocalories from fat (% FAT) = 468.6 x (1 - RQ) 5.047 x (RQ - .707) + 4.686 x (1 - RQ) x KCAL ratio Percentage participation of protein in total O consumption (% PROO2) = VO / VO 2 2un 2 39 Ratio of O participation in FAT+CHO to total VO (O ratio) = (VO - VO ) / VO 2 2 2 2un 2 2 Percentage of participation of CHO in total O consumption (% CHOO2) = (100 x RQ - .707 ) x O2 ratio .293 2 Percentage of participation of FAT in total O consumption (% FATO2) = (100 x 1 - RQ ) x O2 ratio .293 2 Protein metabolism (in grams) (PRO) = VO x % PROO / .94 where .94 = liters of O per gram of PRO 2 2 2 Carbohydrate metabolism (in grams) (CHO) = VO x % CHOO / .81 where .81 = liters of O per gram of CHO 2 2 2 Fat metabolism (in grams) (FAT) = VO2 x % FATO / 1.96 where 1.96 = liters of O per gram of FAT 2 2 Predicted energy expenditure (Harris-Benedict) For men: HBEE = 66.487 + (13.7516 x Weight) + (5.0033 x Height) - (6.7550 x Age) For women: HBEE = 655.0955 + (9.5634 x Weight) + (1.8496 x Height - (4.6756 x Age) The following predicted variables are derived based on information provided in the Subject Data window: Upper limit of normal weight for males (Treadmill and Cycle) = (.79 x height (in.) * 2.54)-60.7 Upper limit of normal weight for females (Cycle) = (.65 x height (in.) * 2.54)-42.8 Upper limit of normal weight for females (Treadmill) = (.79 x height (in.) * 2.54)-68.2) For the following predicted equations for VO , substitute the predicted weight value if the actual weight exceeds the predicted weight unless a specific equation for overweight subjects is listed. 2 Predicted VO (males - normal weight - Cycle) 2 40 = Weight (kg) x (50.72 - 0.372 x Age) Predicted VO (males - normal weight - Treadmill) = Weight (kg) x (56.36 - 0.413 x Age) 2 Predicted VO (females - normal weight - Cycle) = (Weight (kg) x 42.8) x (22.78 - 0.17 x Age) 2 Predicted VO (females - overweight - Cycle) = Height (cm) x (14.81 - 0.11 x Age) 2 Predicted VO (females - normal weight - Treadmill) = Weight (kg) x (44.37 - 0.413 x Age) 2 Predicted Heart Rate = 210 - 0.65 * Age Predicted Ve = MVV Predicted Vt = IC x 1000 41 APPENDIX B COMPUTER REQUIREMENTS • The AEI Metabolic System Software must be used with a Microsoft Windows XP Professional [preferred] or Microsoft Windows 7 32-Bit ONLY Professional operating system computer. A “USB” interface port is required to connect to the Interface Box or control unit. A CD/RW drive or USB Flash Drive is recommended for data storage and transfer. • The VGA graphics and monitor must be capable of displaying and should be set in “1024 x 768” format. • The program supports all printers that are “Microsoft Windows” compatible. A default printer driver must be installed to Print or Preview reports. • The AEI Metabolic System Software is designed to operate on a ‘clean’ standalone computer. The computer should not be connected to a network or to the Internet. Also, there should be no other programs operating on the computer at the same time as the AEI Metabolic System Software. • A laptop or small chassis desktop that fits under the monitor is recommended. • Microsoft Windows should be operating in ‘English (United States)’ as standard formatting. • User must have full administrator rights in Microsoft Windows. 42 APPENDIX C CONSUMABLES / SPARE PARTS Part Number Description 15-2002 15-2003 Breathing Valves Breathing Valve, Large, with Trap, Arm Mtg Breathing Valve, Large, with Trap, Arm Mtg, Insp Port Adaptor 15-1150 15-1160 Moxus Calibration Gas Calibration Gas, E Size, Disposable, 0.03% CO2, 21.00% O2, Bal. N2, Moxus Calibration Gas, E Size, Disposable, 4.00% CO2, 16.00% O2, Bal. N2, Moxus 15-1131 15-1132 Calibration Gas, E Size, Disposable, 6.00% CO2, 40.00% O2, Bal. N2, Calibration Gas, E Size, Disposable,15.00% CO2, 40.00% O2, Bal. N2, 15-2300 Calibration Syringe, 3-Liter (1.375 ID O-Ring) 15-1045 Mask Kit (2) Small and (1) Medium Masks (series 7450), Mask Calipers, Head Cap, Adaptor for breathing valve. Used in place of Mouth Piece. 669197 669196 669195 201492 201493 201494 691143 200525 612738 Masks Parts 669197 Small Mask 114 (7450) 669196 Medium Mask 113 (7450) 669195 Large Mask 116 (7450) 201492 Headgear Small HG-SM 201493 Headgear Medium HG-SM 201494 Headgear Large HG-LRG 691143 Caliper 200525 Head cap 612738 Adapter (Madp 7450) 37082JE 37083JE 37084JE 42276JE 42176JE 42173JE Mouth Piece Parts Mouth piece, Medium Bite, Silicone Rubber Mouth piece, Large Bite, Silicone Rubber Mouth piece, Small Bite, Silicone Rubber Nose clips, Reusable Headgear, Holder for Rudolph Valve Headgear Cover 15-1020 15-1023 12-1031 Dryer Kit Efficient Nafion-based desiccant box with 1 Liter bottle molecular sieve & silica gel indicator Nafion Drying Tube Desiccant, Indicator, 1 Liter 15-1780 Breathing Tubing, Corrugated, 35 mm, 9 ft. 43 15-1781 15-4020 HR- WATCH HR- REC 15-1700 Sample Tubing and Fitting Kit 10 ft of 1/16 inch, and 20 ft of 1/8 inch, 10 male 1/16 in Leur Fittings 10 female 1/16 inch Leur Fittings 10 1/8 inch male Leur Fittings Polar Heart Rate Monitor Heart Monitor System (Monitor Reciever & Cable) Heart Monitor (HR - WATCH) Heart Monitor Reciever (HR - REC) Heart Monitor Cable (15 - 1700) 44 APPENDIX D TROUBLESHOOTING DO I KNOW THAT IT IS WORKING? The first step is to pull down the "Tools" menu, and select "Hardware DX", which displays the Hardware Diagnostics window. MAKE CERTAIN THE CORRECT SYSTEM, FLOW DEVICE, AND OPTIONS ARE SELECTED. This window also allows measuring the voltages of all of the signals coming into the computer, in volts, in digital counts, or in actual calibration values. A good place to start is comparing the data presented on this window in volts with the expected analog voltage output of a device. If this is not the case, the calibration gas being used may not contain the exact value of gas listed on the label. As an example, if a gas is labeled simply as 16% Oxygen but the S-3A/I CONSISTENTLY reads 16.15% (after being set to read ambient air correctly), this may be the accurate measurement of the gas. We would recommend toggling between ambient and the questionable calibration gas to determine the true value of the cylinder. Once again, if the analyzer reads the same value for the gas consistently (16.15%), we would advise you to contact your gas supplier. 45 APPENDIX E CARDIAC OUTPUT MODULE [Optional] CARBON DIOXIDE REBREATHING METHOD SETTING UP OF THE SYSTEM Select Menu: Tools > Hardware Dx Check the Cardiac Output checkbox. Choose Properties/Setup/Cardiac Output. Set the Low CO2-Low O2 and High CO2-High O2 values of the gas cylinders. The Low O2 (regardless of its label) should be the O2 level in the Low CO2 cylinder. The High O2 should be the O2 level in the High CO2 cylinder. Set the target ETCO2 level as the torr level above the end-tidal CO2 during rebreathing. For instance, if the end-tidal CO2 is 40 and a rebreathing CO2 of 46 is desired, set the target rebreathing CO2 level at 6 torr above the end-tidal CO2. Set the Volume Fill for the rebreathing bag as a multiple of the tidal volume. For instance, if the tidal volume is 1.0 liters and you wish to have the bag filled to 1.5 liters, enter a setting of 1.5. This setting will yield a bag volume of 3 liters if the patient’s tidal volume is 2 liters. A setting of 1.5 is initially recommended. Initial studies have shown it to be the minimal volume achievable without complete collapsing of the bag as rebreathing of CO2 progressively triggers an increase in tidal volume throughout the cardiac output test. Max Bag Fill Volume is set by default to 5000 ml. This setting is used primarily for safety to prevent overfilling the rebreathing bag. For a standard 5 liter rebreathing bag, we recommend leaving it set at the default value. # Breaths ignored after CO is set to 10 by default. The purpose of this entry is to allow the subject’s ventilation to reestablish to normal levels following carbon dioxide rebreathing with resulting tachypnea. This, in conjunction with normal analyzer delays and dead space volume settings, accounts for the delay seen in the resumption of data plots following cardiac output studies. Press Save and OK to exit the window. Electrical connections Connect the 9 pin DB ribbon cable between the Cardiac Output module and Cardiac Output connection on the Interface Box [or MAX-II]. Connect the Cardiac Output module to power. Connect BNC ‘T’ connector common to the etCO2 connector on the Interface Box. Disconnect the BNC cable from the CO2 input on the Interface box [cable from the CD3A analyzer] and connect to one end of the BNC ‘T’ connector. Connect the supplied BNC cable from the other end of the BNC ‘T’ connector to the CO2 input on the Interface box. 46 Tubing connections and valve assembly Connect the 15%CO2/40%O2 and 6%CO2/40%O2 cylinders the cardiac output module Hi and Low Fill Tank fittings with 1/8 inch ID tubing, respectively. The outlet pressure on both regulators should be set to approximately 20 psig and resulting in the flow to the bag of about 100 ml./sec. Connect the compressed air cylinder to the system with the white plastic quick connect hose to the Pressure Tank connector. The outlet pressure on this regulator should be set to 55 psig. Remove the mouth port from the standard Hans Rudolph 2700 valve and attach the Hans Rudolph cardiac output valve in it’s place as shown below: Breathing Valve Breathing Valve is shown in blue. Cardiac output valve is shown in orange. The cardiac output valve and bag assembly is supplied with tubing connected at one end. Connect the other end to the rear of the Cardiac Output module as follows: Connect small 1/16 inch ID tubing to the Mouth Port fitting . Connect large 1/4 inch ID tubing to the Empty Bag fitting. Connect 1/8 inch ID tubing with Black tape to the Top Balloon fitting. Connect 1/8 inch ID tubing with Blue tape to the Bottom Balloon fitting . Connect 1 of the 2 remaining 1/8 inch ID tubing to the Fill Bag fitting. Connect the remaining 1/8 inch ID tubing to the Sensing fitting. Disconnect the Nafion tube from the Interface Box and connect to the Dryer port on the Cardiac Output Module using 1/16 inch ID tubing. Keep the tube as short as possible. 47 Connect from the Dryer top barb on the Interface Box to the Cal Valve port on the Cardiac Output Module using 1/16 inch ID tubing. Keep the tube as short as possible. Testing Equipment Operation We recommend checking all equipment operation prior to the beginning of the test. The Properties window under Tools displays a Cardiac Output tab that allows alteration of settings before and during the test. It also displays a Control tab that allows one to manually operate the equipment, before a test, as shown below: When “Normal” is selected, the lower balloon (the balloon nearest the rebreathing bag) inflates and the upper balloon deflates. The lower balloon should be examined to make sure that it remains fully inflated and does not slowly deflate. When “Rebreathe” is selected, the lower balloon deflates and the upper balloon inflates. The upper balloon should be examined to make sure that it remains fully inflated and does not slowly deflate. If either balloon appears to be “sucked-in”, simply remove and replace the tubing to that balloon, and switch to the other mode (either “normal” or “rebreathe”), to allow air to be sucked into the balloon pump with the disconnected tubing.. When “Fill Bag” is selected, the lower balloon inflates and the rebreathing bag fills, using a 50/50 mixture, to the minimum volume, as indicated by both Hi Fill and Lo Fill lights on the Cardiac Output module illuminating. If the bag does not fill, check all tubing connections, the lower balloon inflation and the pressure setting on the regulators and the cylinder contents. 48 When “Empty Bag” is selected, the Cardiac Output bag evacuation pump activates, and remains “on” until the bag is deflated completely. The pump turns off once the bag is fully deflated. If the pump does not turn off, check the tubing between the vacuum / pressure switch, check the bag for leaks and check the inflation of the lower balloon. All other routine configuration settings are made in the Properties window: Setup tab The O2 and CO2 delays will need to be increased to account for the increased tubing length for the etCO2. Typically, the delays will increase by 2 seconds. Cardiac Output tab The filling of the rebreathing bag with respect to volume and CO2 concentration is automated and changes to meet the patient’s condition. Twenty tidal volumes prior to a CO2 rebreathing trial are observed and averaged. This tidal volume average determines the volume to which the bag will be filled and is influenced by the settings in the “C/O Bag fill volume”. As shown above, the bag will be filled to 3 liters (2 x the average tidal volume) if the subject’s tidal volume is 1.5 liters. Regardless, the bag volume will never be less than 1500 ml nor larger than 5000 ml regardless of the tidal volume as a safety measure. As the filling of the bag is based on time, the software needs to know the filling flow rate of the gas mixture. Typically, this will be about 100 ml/sec if the gas regulators on the cylinders are set to 20 PSIG. The target bag CO2 concentration is achieved by proportionally mixing gases from both cylinders. The software requires the entry of gas concentrations from both cylinders in 49 order to determine how much of each gas is needed to achieve the proper CO2 target value. It is important that cylinder content values be precisely entered. Therefore, whenever a cylinder is changed, the technologist must examine the cylinder label to ascertain it’s true gas mixture. The target bag CO2 value is determined similarly to the target tidal volume. The software averages 10 CO2 measurements prior to the cardiac output trial. This average is then compared to the value entered under “etCO2 Fill (over average Torr). Therefore, if an average end-tidal CO2 is measured at 40 torr, the software will fill the bag to a concentration of 70 torr (40 torr end-tidal CO2 + setting of 30 torr). Normal oxygen uptake and carbon dioxide production measurements are interrupted while a CO2 rebreathing trial is underway. Upon completion of a cardiac output trial, the extra CO2 that has accumulated in the blood during the rebreathing trial must be washed out of the lungs prior to resumption of VCO2 measurements. Otherwise, spuriously high VCO2 and RER data will result. It is for this reason that there is a setting in the C/O setup window for “# breaths to ignore after CO”. Once all configuration settings are made, click on OK. All settings can be changed during an exercise test to allow modifications for bag filling volumes and target CO2. EtCO2 CALIBRATION Enter the Cal High and Cal Low values for the etCO2 channel on the System Calibration window. These values should be identical to the values for the CO2 channel. Then select both the CO2 and etCO2 channels for calibration. Perform both Hi and Lo software calibration. Save the result. Equilibration point PetCO2 Time EQUATIONS The following equations are used in the program to calculate cardiac output CaCO2 ml = antilog [(loge PaCO2 x 0.396) + 2.38] Note: If entered into the program, arterial CO2 substitutes for PetCO2. CvCO2 ml = antilog [(loge PvCO2 x 0.396) + 2.38] 50 Note: Ven PCO2 is measured by moving the line to the equilibration point on the graph. Cardiac Output (Q) ml/min = VCO2 / ((CvCO2 - CaCO2) x 1000) NOTE: To filter the effect of aberrant breaths on the data, the program averages 20 PetCO2 values and 10 VCO2 values prior to each measurement. 51 Cardiac output has traditionally been measured by the Fick technique which relates the oxygen uptake to the arterial to mixed venous O2 content in the following equation Qt = VO2 / CaO2 – CvO2 Where: Qt = Cardiac Output VO2 = Oxygen Uptake CaO2 = Arterial blood oxygen content CvO2 = Mixed venous blood oxygen content As the collection of arterial and mixed venous blood samples requires the invasive placement of catheters, this technique has not enjoyed widespread use in exercise physiology measurements. An indirect Fick method has been developed which substitutes the use of CO2 measurements in the equation as follows: Qt = VCO2 / CaCO2 – CvCO2 Where: VCO2 = Carbon dioxide production CaCO2 = Arterial blood carbon dioxide content CvCO2 = Mixed venous blood carbon dioxide content Arterial blood CO2 content is non-invasively estimated by measuring the expired CO2 at the mouth with a rapid CO2 analyzer and examining the observed waveform for it’s peak. This measurement is commonly referred to as “end-tidal CO2 or PetCO2” and is measured in mmHg [torr]. This measurement closely approximates PaCO2 (the partial pressure of CO2 in arterial blood) in subjects with normal lungs. CaCO2 is then calculated with the following formula: CaCO2 (ml) = antilog [(loge PaCO2 x 0.296) + 2.38] For subjects with underlying pulmonary disease, the software allows entry of arterial blood PCO2 in place of end-tidal PCO2 measurements in order to more accurately estimate cardiac output. Measurement of VCO2, of course, is non-invasive and is routinely measured during exercise testing. The measurement of mixed-venous CO2 content can also be approximated non-invasively. It is known that, if a normal subject rebreathes from a bag filled with CO2 roughly approximating that of the mixed CO2 in the blood, a rapid equilibration occurs between the lungs, the alveolar capillary blood and the rebreathing bag. To be accurate, the equilibration needs to be completed with 10 seconds; otherwise, recirculation of the blood from the lungs through the system and back to the lungs is likely to occur. Thereafter, the result will then be a constant increase of CO2 in the rebreathing bag, nullifying the measurement. This is easily corrected in subsequent measurements by either adjusting the rebreathing bag target CO2 either up or down. The example windows below show acceptable and suboptimal CO2 rebreathing curves. 52 Acceptable CO2 curve showing equilibration at the placement of the blue line CO2 concentration does not equilibrate and continues to rise. CO2 concentration in the bag is too low and needs to be increased on the next measurement. 53 CO2 concentration does not equilibrate and continues to oscillate after 10 seconds. CO2 concentration in the bag is too high and needs to be decreased on the next measurement. SOURCES OF ERROR AND LIMITATIONS WITH THE FICK REBREATHING TECHNIQUE The Fick method of measures cardiac output by an indirect method. As with any indirect measurement, the accuracy of the measurement is subject to certain conditions. The "Fick" method of measuring cardiac output is based on the assumption that the cardiac output of an individual is the result of dividing the CO2 production of that individual by the difference between the arterial CO2 (coming out of the lungs) and the mixed venous CO2 (going into the lungs). To do this measurement accurately, we must, first, measure the mixed expired CO2 and the end-tidal CO2, immediately before rebreathing. This is a valid assumption if the subject is at a constant level of activity during these measurements. Another assumption is that, if a subject rebreathes his or her exhaled gas, under certain circumstances, that an equilibration point will be reached which is equal to the CO2 concentration of mixed venous blood. This may be valid when the test subject has healthy lungs, and when the tidal volume of the subject is large enough to flush out his or her dead space (bronchi, pharynx, etc.). In subjects with impaired breathing, and significant intrapulmonary mixing, or in subjects with very low tidal volumes (less than 2x dead-space), the "Fick" method determines the mixed venous value inaccurately. Another basic assumption, is that the end-tidal CO2 partial pressure, represents a similar value to the alveolar CO2 partial pressure. This also is only true for subjects with healthy, homogeneous, lungs, and it is not true for those with lung disease, or those who breathe with very low tidal volumes (insufficient to wash out their dead space). 54 Another assumption, that must be made, is that the additional CO2 that is generated in the rebreathing process will not return to the lungs before equilibration is reached. It is generally assumed that if equilibration occurs within 10 seconds of the onset of rebreathing, that this will not be a factor. In order to ensure rapid equilibration the CO2 content and the volume added to the rebreathing bag is critical to hasten equilibration. The analysis of the gas in the rebreathing bag is, also, not without its problems. Background levels of oxygen, within the bag, affect the measurement of CO2 by nondispersive infrared analyzers. To be assured that the test subject does not become hypoxic, while rebreathing, the bag must be filled with a higher concentration of oxygen than room air (typically 40%), which is reduced as the subject consumes oxygen during rebreathing. The changing level of oxygen during rebreathing has an effect on the measurement of the CO2 concentration; however, it has minimal error [<2%]. Higher levels of CO2 are a stimulant to respiration. Because of this, the "Fick" rebreathing procedure is very uncomfortable at higher levels of exercise. This test is much more tolerable at lower levels of exercise, or at rest. RUNNING A CARDIAC OUTPUT TEST Prior to running a test flow volume and all analyzers must be calibrated. See the Calibration section for further instructions. Once the test has begun and after a 10 breath waiting period the Cardiac Output button appears. Pressing this button will enable the Cardiac Output process. A Cardiac Output status box will appear. The rebreathing bag will initially empty at the start of the program to begin preparing it for the next cardiac output trial. The next status box shows Measuring CO2. During this time, the system is measuring the averaged VCO2 used in cardiac output calculations. The system is performing the standard metabolic measurements until the Fill Bag button is pressed. The window shows the CO2 overfill – The amount of CO2 in torr to add to the measured end-tidal CO2 to determine the target value (see above). This can be changed before each CO measurement. NOTE: as workloads are changed the subject should be allowed to stabilize at the workload prior to starting a cardiac output measurement. It is advised to note the CO2 and Volume reading just before the initiation of cardiac output. Following the CO measurement, the subject should be allowed to reestablish these measurements prior to calling additional measurements in order to assure that all rebreathed CO2 has been cleared from the subject and normal VCO2 is achieved. 55 Cardiac output testing is begun by clicking on “Fill Bag” button. The rebreathing bag is filled (Fills bag to the target CO2 concentration and volume. Note that the minimum and maximum volume are always as set in the CO Setup window under Properties. Therefore, if the minimum volume was set at 1500 ml, then the bag will be filled to 1500, even if the Vt multiplier results in a volume less than this minimum volume. The same rule applies to maximum volume.) The system will now wait for 20 processed breaths to occur to determine the averaged tidal volume and VCO2. NOTE: It is important to remember that the subject is rebreathing CO2 and will experience dizziness, shortness of breath and tachycardia if the test is prolonged more than necessary. The test should be terminated as soon as equilibration is achieved (shown by the arrow), ideally within a 10 second window and, more importantly, before recirculation begins. NOTE: For safety reasons, the test will automatically end and the subject will be switched back to normal metabolic measurement if the test is prolonged up to 30 seconds. This is immediately followed by the re-breathing process. The picture shows the cardiac output rebreathing display. Allow the patient to rebreathe from the bag until an equilibration is achieved. Click DONE when the subject has reached equilibration or you have determined that equilibration will not be achieved at this time. The Cardiac Output test will terminate and the normal testing sequence will continue. However, this window will remain open to allow setting the equilibration point manually via the mouse. Click OK to close this window and display the Cardiac Output value. Following a cardiac output test, the status box will sequence through Emptying Bag and Ignoring Breaths (To allow washout of the extra CO2 loaded to the patient during the rebreathing trial.) 56 NORE: The tabular metabolic data should be observed for restabilization of the target volume and CO2 before another cardiac output trial is measured. This assures that all rebreathed CO2 is cleared from the subject and that the RER is stabilized. If equilibration is not acceptable adjust the etCO2 Overfill amount in the window before starting another CO test. Typically this adjustment is no more than +/- 3 Torr. Additional trials can be measured simply by clicking on the “Fill Bag” button. 57 After the last trial, it is recommended that the tabular metabolic data be allowed to restabilize before stopping the test and removing the subject from the mouthpiece. This will allow the phase delays from normal metabolic measurements prior to the cardiac output trial to be processed and the CO data to be properly tagged in time. Following completion of the metabolic test, the cardiac output trials must be reviewed and processed for the data to appear on the report. PROCESSING CARDIAC OUTPUT DATA First press the Preview CO button. Processing of data consists of examining the equilibration curve from each of the trials, deciding whether or not the equilibration is valid, rejecting those that are deemed invalid, identifying the equilibration point, adjusting the equilibration marker as needed, and manually entering PaCO2 when it is directly measured from arterial blood. A typical processing window for CO2 equilibration is displayed on the next page: Detailed instructions are provided in the window located in the lower left hand corner. The red downward sloping straight line displayed on the graph represents an estimate of the FiO2 in the rebreathing bag based on the oxygen consumption just prior to the 58 beginning of the trial and the starting FiO2 in the bag. Time, Breath number and Cardiac Output Sequence (trial number) are displayed to assist the operator in determining the timing of the measurement relative to the entire exercise test. Upon viewing a graph, the technologist should first determine whether or not the curve is valid for measurement. There should be a discernable equilibration point where there is minimal to no oscillation of the CO2 tracing and it must occur within ten seconds from the onset of rebreathing (the time at which the CO2 tracing rises from the baseline to start the equilibration process). If no discernable equilibration is present, the trial should be deemed invalid, and the checkmark should be removed from the “Data Valid” checkbox. Next, the technologist should determine whether or not he/she agrees with the computer’s determination of the equilibration point. If not, the equilibration line is easily moved using the left button mouse drag function. Both the Venous pCO2 and Cardiac Output readings will instantly change as the line is dragged to the desired placement point. Normally, the “Arterial pCO2” value, which is displayed, represents the averaged endtidal CO2 measured just before the trial. If desired, a measured arterial blood PCO2 59 result may be manually entered in substitution. The Cardiac Output reading will be updated once the mouse is clicked outside of this box. Additional trials may be evaluated by either clicking the Next button or clicking on the Up arrow next to the Cardiac Output Sequence window. At any point, you may view earlier trials by clicking on the Down arrow next to the Cardiac Output Sequence window or clicking on the Previous button. The Cancel button exits from the measurement and disregards all changes. Once all graphs have been processed, click on the OK button to store the data. VIEWING GRAPHS AND PRINTING REPORTS Once the data is processed and selected, cardiac output (CO) and Venous PCO2 data is automatically display in the window and printed tabular data listing. Cardiac output and mixed venous PCO2 data is also available for graphing against time or any other test parameter. 60 APPENDIX F CANOPY SYSTEM ***CAUTION*** Constant attention to the subject by a qualified individual is required at all times when the subject is under the canopy. If air flow through the canopy stops or a loss of power occurs immediately remove the canopy from the subject. NOTE: The following steps, procedures, and features apply only to the Canopy System Option. Canopy System Setup • • • • • • • • Place the Canopy Pump assembly on the bottom shelf of the cart and plug power cord into the power strip on the cart. Remove thin plastic protective cover, if any, from the Canopy hood. Attach the plastic Drape to the Velcro on the outer lip of the Canopy Hood. Attach a Canopy Connector each end of the Canopy Hood. The connector will unscrew into 2 parts; then fit through the hood and screw parts together. Connect the supplied 7/8” corrugated hose to the Pneumotach using the supplied connector [white plastic piece about fist sized]. Connect the other end of the 7/8” corrugated hose to the Canopy Connector at the NARROW end of the hood. This end of the hood is closest to the mouth. A short 30cm piece of the 7/8” corrugated hose can be attached to the other connector in situations where people may introduce exhaled breath to the hood’s inlet port. Connect a large corrugated hose from the other end of the Pneumotach to the Mixing Chamber input. Change the 3-way Valve on the side of the Canopy Pump to the Vertical position. Software Setup RUN AEI METABOLIC SYSTEMS SOFTWARE PROGRAM In addition to the Setup of the Software perform the following: 61 Select Menu: Tools > Hardware Dx • Select: CANOPY • Click OK • The program will re-initialize. Completely close the program. Re-start the program. Volume [Flow] Calibration for Canopy Option • • • • Disconnect the hose from the expiratory side of the Pneumotach [where it connects to the Mixing Chamber]. Connect another hose to the inspiratory side of the breathing valve and the expiratory side of the Pneumotach. Perform Flow Calibration as described on Page 12 using a stroke rate of 7-8 seconds for each inspiration. When Flow calibration and validation is completed re-connect the hose from the Mixing Chamber to the expiratory side of the Pneumotach. Running a Test • • After entering Subject information and starting the Canopy Pump place the Canopy Hood over the Subject’s head. Adjust the Hood location such that the subject’s mouth is about 10cm away from the outlet of the Hood and then tuck the drape under the subject. A small pillow under the subject’s head may be desirable for comfort. The Canopy System continuously monitors and displays the flow rate of air passing through the canopy in Liters/Minute. Adjust the Flow Rate on the Real Time window such that the feCO2 is in the desired range [typically 0.7 – 0.8 %CO2]. The Flow Rate is adjusted by SLOWLY turning the black knob on the top of the large Canopy Pump [gray metal box on the bottom shelf]. Clockwise rotation of the knob decreases flow; counter-clockwise rotation of the knob increases flow. NOTE: Do not turn the knob to completely shut off air flow through the canopy. Converting Canopy System to Exercise System • Select Menu: Tools > Hardware Dx • De-Select: CANOPY • Click OK • The program will re-initialize. Completely close the program. Re-start the program. • Disconnect the tubes and adaptors from the Pneumotach. • Connect the Pneumotach to the inspiratory port of the breathing valve with a large corrugated hose. • Connect the large corrugated hose from the Mixing Chamber input to the expiratory port of the breathing valve. • Turn the 3-way Valve mounted on the Canopy Pump to the Horizontal position. • To revert back to a Canopy System, reverse the above steps. 62 APPENDIX G CLINICAL SYSTEM OPERATION NOTE: Operating in the Clinical System mode does not imply that the MOXUS Metabolic System is approved for operation as a medical device. Please contact AEI Technologies for details. The MOXUS Metabolic System has an optional mode that allows use of Clinical System features such as Data Encryption, Password Protection, and Subject Name ID. To enable the Clinical System operation: Select Menu: Tools > Hardware Dx • Select: CLINICAL SYSTEM • Click OK • The program will re-initialize. Completely close the program. Re-start the program. To disable the Clinical System operation: NOTE: you need to use the ‘admin’ password [found in the Installation Instructions] to disable this feature. Select Menu: Tools > Hardware Dx • De-select: CLINICAL SYSTEM • Click OK • The program will re-initialize. Completely close the program. Re-start the program. Data Encryption In Clinical System operation all data will be encrypted utilizing the TrueCrypt encryption freeware. The TrueCrypt program and associated documentation is available for free download from their website: www.truecrypt.org. To install and setup TrueCrypt please refer to the documentation available on their website or contact your network administrator. The Volume File created is then Mounted as a virtual Drive m:. All data is then encrypted and stored in Drive m:; including all *.dat data files, all *.xls files exported to Excel, and the REErpt.txt report text file. Password Protection In Clinical System operation a Password is required to operate the Metabolic System software. There will be 2 levels of password protection: User and Admin. The User password is simply a null [just click OK; do not type any characters]. This will allow 63 Calibration, running a Test, most Features, printing and viewing Reports. Access to alter Setup information [Hardware Diagnostics, Properties, and Preferences windows] is not allowed. The Admin password is found in the Installation Instructions and allows complete access to all commands and features of the software. Subject Name ID In Clinical System operation the subject’s name should not be entered on the Subject Data window. Please enter a Subject ID instead. The data file will then contain the Subject ID, Date and Time rather than the Subject Name, Date and Time. AEI Technologies, Inc. 201 Hunters Crossing Blvd Suite 10-171 Bastrop, TX 78602 800-793-7751 630-548-3545 Fax: 630-548-3546 Sales@aeitechnologies.com www.aeitechnologies.com 64
© Copyright 2025